Understanding Time in Range helps manage diabetes effectively. Learn how CGM metrics reveal hidden patterns for better health outcomes.
Archive: 2026/03
Learn about convergence insufficiency therapy, diagnosis methods, and treatment effectiveness for binocular vision disorders affecting eye coordination.
Understand school medication safety rules including forms, drop-off procedures, and self-administration policies to ensure your child’s health is protected daily.
Medication errors harm millions each year - but most are preventable. Learn how hospitals use barcode scanning and AI to cut mistakes, and what you can do at home to stay safe.
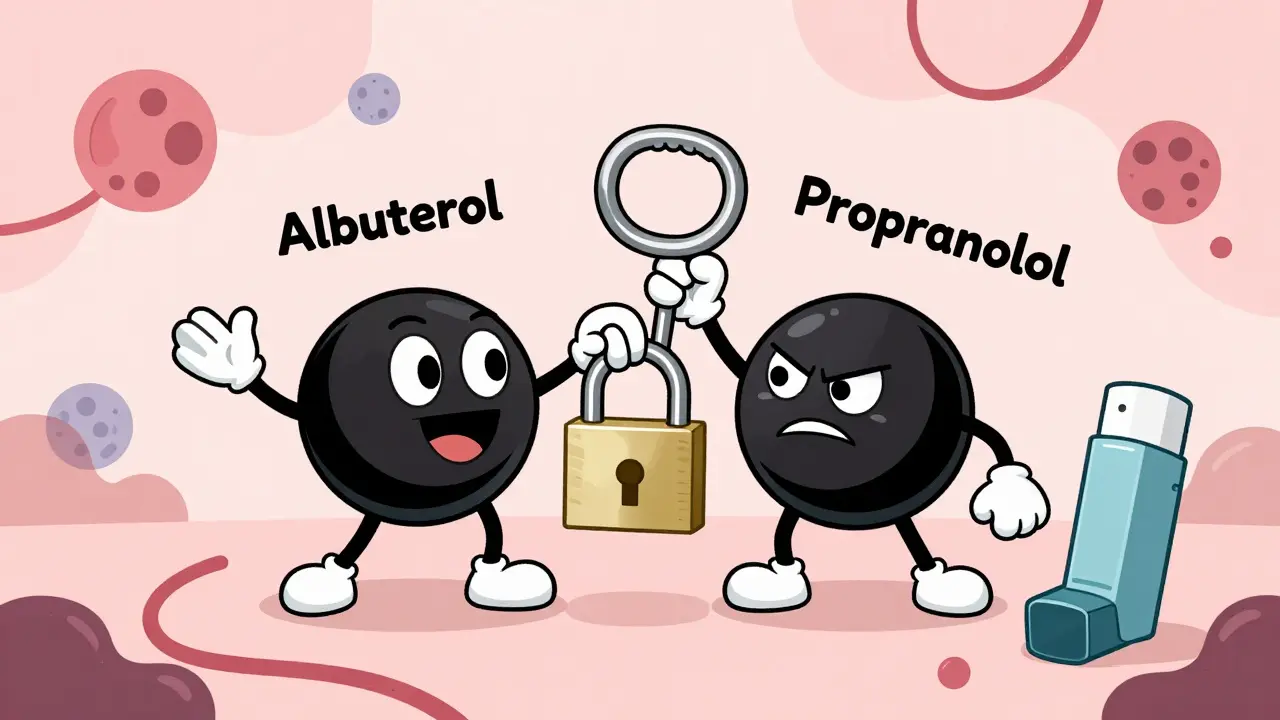
Pharmacodynamic drug interactions occur when drugs alter each other's effects at the receptor level-not by changing blood levels, but by changing how they work. These hidden interactions can be deadly or lifesaving, and are responsible for 40% of serious drug-related hospitalizations.
First generics and authorized generics both offer cheaper versions of brand-name drugs, but their timing and strategy differ dramatically. Authorized generics, launched by the original brand company, often enter the market on the same day as the first generic - slashing profits and limiting price drops. This tactic undermines the incentives built into the Hatch-Waxman Act.
Itching from Medications: Causes and What You Can Do
 Mar, 12 2026
Mar, 12 2026
Itching from medications is a real and often overlooked side effect. Learn which drugs cause it, why antihistamines sometimes make it worse, and what actually works to treat it.
Global substitution laws like the UK's GSO let companies transfer thousands of legal claims with one court order, cutting costs by up to 85%. Learn how it works, where it's used, and why it's changing international debt recovery.
Replicate study designs are essential for assessing bioequivalence in highly variable drugs, reducing sample sizes by up to 75% while maintaining statistical rigor. Learn how full and partial designs work, regulatory differences between FDA and EMA, and why they’re now the industry standard.
Generic drug manufacturing standards use strict quality controls to prevent counterfeit medicines. From cGMP rules to digital tracking, this system ensures every pill is safe, effective, and authentic.
Antispasmodics like dicyclomine and hyoscine can cause dangerous interactions when combined with common medications. Learn which drugs to avoid, who’s at risk, and how to stay safe.
How to Document Safety Alerts on Your Medication List: A Practical Guide for Healthcare Teams
 Mar, 1 2026
Mar, 1 2026
Documenting safety alerts on medication lists prevents deadly errors. Learn how to implement ISMP guidelines, track high-alert drugs like insulin and anticoagulants, avoid alert fatigue, and build a system that actually works - not just paperwork.